Sleep and Spinal Cord Stimulation After Failed Back Surgery
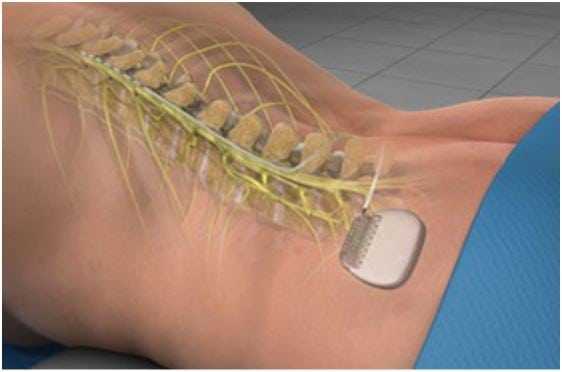
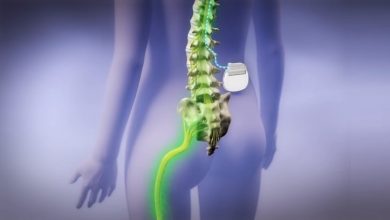
Spinal cord stimulators may be used for pain management in people with Failed Back Surgery Syndrome but one potential benefit of these devices has largely been overlooked, until now. Details of new research were published in Neurochirurgie (Neurosurgery) last month and describe a pilot study aiming to investigate the effects of spinal cord stimulation on sleep, something which is frequently impaired in people with chronic pain, including pain originating from Failed Back Surgery Syndrome (FBSS).
FBSS can occur for a variety of reasons and may involve symptoms such as persistent back pain, leg pain, radiculopathy, numbness, weakness, and other symptoms. One majory effect of FBSS is a significant reduction in the quality of sleep. Poor sleep quality and chronic pain are both linked to reduced quality of life and psychiatric problems including depression and even suicide.
New Study for SCS and Sleep Underway
As spinal cord stimulation (SCS) has been the subject of much recent study for its use in treating FBSS, researchers Monlezun and colleagues decided to assess the effect of SCS on patients’ sleep quality. This pilot study is set to take place at a single centre in France and will involve 16 FBSS patients scheduled to undergo implantation with multicolumn SCS. These patients have now been recruited and enrolled in the French multicentre ESTIMET study and will be monitored for 6 months after implantation to determine the effects, if any, of spinal cord stimulation after failed back surgery.
The researchers will assess a variety of parameters of sleep quality through the use of polysomnography, Psychomotor Vigilance Test and Osler tests, actigraphy, sleepiness scales, and sleep quality testing. Patients will be tested at baseline (before the SCS devices are implanted) and will be reassessed at the 6-month follow-up. The researchers will also look at the effect of SCS lead programming, specifically any differences between mono and multicolumn SCS, and at the effect on sleep of position-adaptive stimulation at night.
Could SCS Help Improve Quality of Sleep, and Life, in FBSS?
Results of this trial are expected in 2015, with the first patient recruited back in 2012. The hope is that patients who are being considered for implantation with an SCS device for FBSS will also, eventually, be assessed for sleep quality as part of the decision-making process, should the results of this study indicate a clear benefit for this symptoms of FBSS. Many patients suffering with FBSS end up taking numerous medications, while an SCS device could help reduce reliance on such drugs and reduce their risk of side effects.
SCS devices are not, of course, without their own safety concerns. Indeed, recent research indicated that, in order to reduce the risks associated with such devices, SCS implantations should always be carried out by a qualified and experienced surgeon at an appropriate facility. The short-term cost of an SCS may prevent many patients from accessing such therapy for FBSS but it may be that further understanding of their long-term benefit, including for sleep quality and associated morbidities, could help tip the scale when deciding on how best to treat this complex result of failed back surgery.